What is Hepatic Lipase Deficiency (HLD)?
Hepatic Lipase Deficiency (HLD) is a rare genetic condition that usually presents with dyslipidemia, or increased levels of triacylglycerides or cholesterol in the blood. It is associated with an increased risk of developing coronary artery disease (atherosclerosis) and heart disease. Mutations, or changes, in the LIPC gene lead to the development of the condition. These changes can be transmitted in an autosomal recessive manner [1].
Symptoms of HLD
Hepatic lipase deficiency is normally characterized by increased levels of triglycerides and cholesterol in the blood. Affected persons may also display increased levels of high-density lipoproteins (HDLs) and decreased levels of low-density lipoproteins (LDLs). Diagnosis is based on many factors including, but not limited to, family history, patient medical history, symptoms, and laboratory testing. If you have questions about diagnosis please contact a medical professional [2].
Hepatic lipase deficiency is normally characterized by increased levels of triglycerides and cholesterol in the blood. Affected persons may also display increased levels of high-density lipoproteins (HDLs) and decreased levels of low-density lipoproteins (LDLs). Diagnosis is based on many factors including, but not limited to, family history, patient medical history, symptoms, and laboratory testing. If you have questions about diagnosis please contact a medical professional [2].
Lipid |
Normal Levels |
Borderline High Levels |
High Levels |
Triglycerides |
<150 mg/dL |
150 - 199 mg/dL |
>500 mg/dL |
Cholesterol |
<200 mg/dL |
200 - 239 mg/dL |
>240 mg/dL |
Table 1. Desirable lipid levels in adult humans [3, 4].
What causes HLD?
Lipase C (LIPC) gene
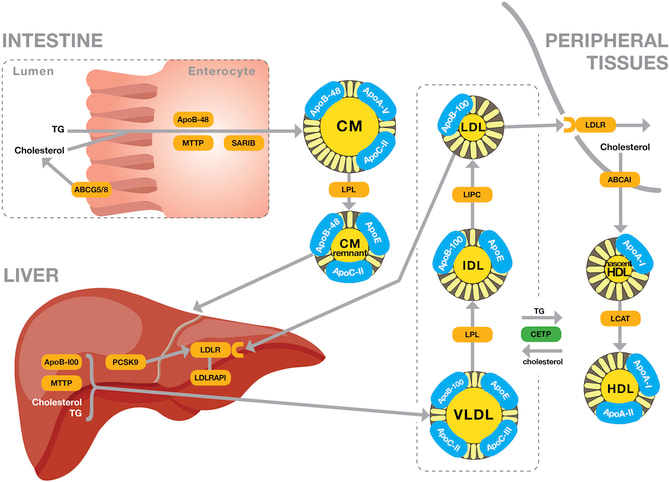
Mutations in Lipase C lead to the development of hepatic lipase deficiency. LIPC encodes instructions to make the enzyme hepatic lipase (HL). Hepatic lipase normally converts very low-density lipoproteins (VLDLs) and intermediate-density lipoproteins (IDLs) into low-density lipoproteins (LDLs) (Figure 1). It also helps transport high-density lipoproteins (HDLs) carrying cholesterol and triglycerides from the blood into the liver. Once in the liver, cholesterol and triglycerides are redistributed or removed from the body. In order to carry out these functions, hepatic lipase must be produced by liver cells and then secreted into the bloodstream. Mutations that lead to the development of HLD either prevent the release of hepatic lipase form the liver or decreases its enzymatic activity once in the bloodstream. This alters the lipid levels in an affected person’s bloodstream, causing abnormally high cholesterol and triglyceride levels [5].
Mutations in Lipase C lead to the development of hepatic lipase deficiency. LIPC encodes instructions to make the enzyme hepatic lipase (HL). Hepatic lipase normally converts very low-density lipoproteins (VLDLs) and intermediate-density lipoproteins (IDLs) into low-density lipoproteins (LDLs) (Figure 1). It also helps transport high-density lipoproteins (HDLs) carrying cholesterol and triglycerides from the blood into the liver. Once in the liver, cholesterol and triglycerides are redistributed or removed from the body. In order to carry out these functions, hepatic lipase must be produced by liver cells and then secreted into the bloodstream. Mutations that lead to the development of HLD either prevent the release of hepatic lipase form the liver or decreases its enzymatic activity once in the bloodstream. This alters the lipid levels in an affected person’s bloodstream, causing abnormally high cholesterol and triglyceride levels [5].
What are the outcomes of HLD?
|
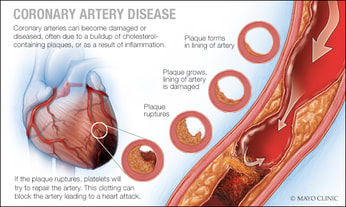
Coronary Artery Disease (CAD)
Hepatic lipase deficiency may be associated with an increased risk of developing coronary artery disease (CAD or atheroschlerosis). However, more research is needed to determine the long term effects of elevated triglycerides and cholesterol in the blood [2, 5]. |
What model organism can be used to study HLD's effects on reproduction?

Rat
Rattus norvegicus
This was the first mammalian species domesticated for scientific research, with work dating back to before 1850. The rat has become the most widely studied experimental animal model for biomedical research [7].
Rat database
Rattus norvegicus
This was the first mammalian species domesticated for scientific research, with work dating back to before 1850. The rat has become the most widely studied experimental animal model for biomedical research [7].
Rat database
Rattus norvegicus is a good model for studying the effects of hepatic lipase deficiency on reproduction as rats ovaries are more similar to humans than those of mice. In addition, they share a progesterone synthesis pathway that is highly similar to humans [8].
What are the goals of this project?
The long-term goal of this project is to identify how LIPC is involved in reproduction to improve infertility treatments for patients with hepatic lipase deficiency. The goal of this specific project is to identify how LIPC isoforms interact to promote cholesterol substrate uptake. My hypothesis is that the extracellular form of LIPC promotes remodeling of intermediate-density lipoproteins into low-density lipoproteins that are taken up by the granulosa cells while the intracellular truncated isoform of LIPC mobilizes cholesteryl esters from lipid droplets to free the cholesterol for the synthesis of progesterone and other reproductive hormones. Rattus norvegicus will be used as a model organism due to its similar progesterone synthesis mechanism to humans and the presence of a close ortholog to human LIPC [9, 10].
Project Aims
AIM 1: Investigate the effects of 3 glycosylated residues in exons one and two of full-length hepatic lipase.
Approach: Domain analysis has revealed the existence of three conserved glycosylated residues in exons one and two of LIPC. To investigate the purpose of these residues, I will mutagenize them using CRISPR-Cas9 to prevent their glycosylation. The effects of these changes on LIPC activity and localization can then be measured through triglyceride lipase activity assays and fluorescence microscopy.
Rationale: Glycosylation is important for the function of secreted proteins. The presence of these conserved residues in only the full-length LIPC isoform suggest that they are important for extracellular LIPC function.
Hypothesis: Mutagenesis of these conserved glycosylation residues will reduce LIPC function and potentially inhibit secretion. If these sites are the drivers for full-length LIPC function, I may see a complete switch to the truncated LIPC isoform activity.
Rationale: Glycosylation is important for the function of secreted proteins. The presence of these conserved residues in only the full-length LIPC isoform suggest that they are important for extracellular LIPC function.
Hypothesis: Mutagenesis of these conserved glycosylation residues will reduce LIPC function and potentially inhibit secretion. If these sites are the drivers for full-length LIPC function, I may see a complete switch to the truncated LIPC isoform activity.
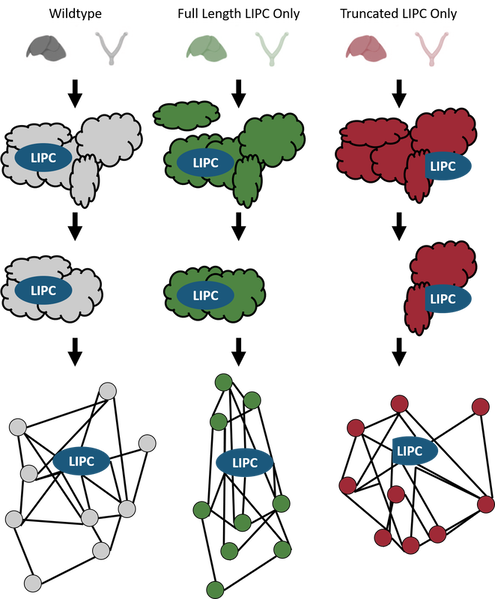
AIM 2: Characterize differentially expressed genes in rats that only express truncated LIPC or full-length LIPC.
|
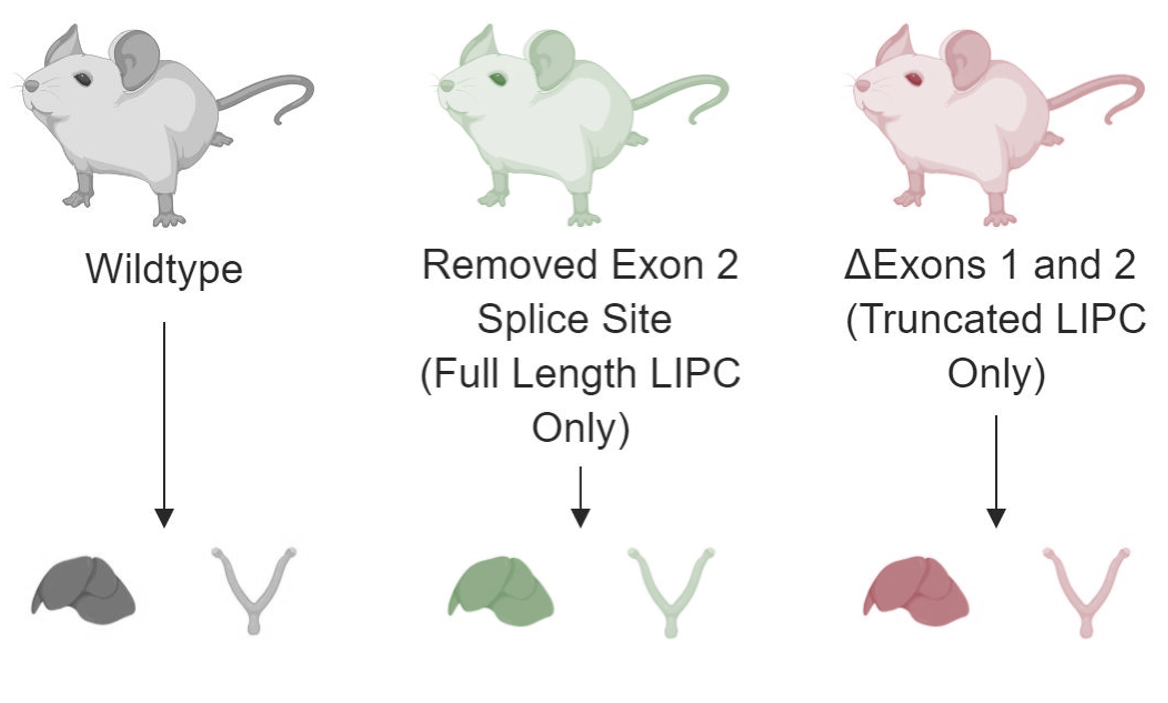
Approach: I will create two mutant rat lines. One will have a mutation in the exon two splice site that prevents the truncated version of LIPC from being created. In other words, this line will only express full-length LIPC. The other line will have a deletion of exons one and two, meaning all the LIPC it expresses will be in the truncated isoform. Once these lines are created, I will extract the livers and ovaries from each line for RNA-sequencing. I will then compare the results to those of a wildtype mouse line to detect changes in gene expression between wildtype mice and the mutants. Genes with differential expression will be subject to gene ontology analysis to determine pathways that are affected by the presence of only full-length LIPC or only truncated LIPC.
|
Rationale: RNA sequencing will determine differential gene expression of genes that are affected by the pathways involving both full-length and truncated LIPC. Gene ontology analysis can further pin down pathways involved in LIPC function.
Hypothesis: I expect to see differential expression of genes involved in lipoprotein remodeling in the mutant line that only expresses truncated LIPC. Likewise, for the line that only expresses full-length LIPC I would expect to see differential expression of genes involved in lipid droplet remodeling
Hypothesis: I expect to see differential expression of genes involved in lipoprotein remodeling in the mutant line that only expresses truncated LIPC. Likewise, for the line that only expresses full-length LIPC I would expect to see differential expression of genes involved in lipid droplet remodeling
AIM 3: Identifying protein interactions of both truncated and full-length LIPC isoforms
Approach: The truncated-only and the full-length-only LIPC mutant rat lines will be used to study the protein interactions of each isoform. I will make lysates from the liver and ovaries of rats from each mutant line and perform a tandem-affinity purification (TAP) against LIPC. The resulting samples will be run through mass spectrometry analysis to identify proteins interacting with truncated or full-length LIPC.
Rationale: Performing TAP and mass spectrometry will provide direct evidence for the proteins that interact with the full-length and truncated isoforms of LIPC.
Hypothesis: I expect that proteins involved with lipoprotein and chylomicron remodeling interact with the full-length LIPC isoform while proteins involved in lipid droplet remodeling interact with the truncated isoform of LIPC.
Rationale: Performing TAP and mass spectrometry will provide direct evidence for the proteins that interact with the full-length and truncated isoforms of LIPC.
Hypothesis: I expect that proteins involved with lipoprotein and chylomicron remodeling interact with the full-length LIPC isoform while proteins involved in lipid droplet remodeling interact with the truncated isoform of LIPC.
References
[1] Connelly, P. W., & Hegele, R. A. (1998). Hepatic Lipase Deficiency. Critical Reviews in Clinical Laboratory Sciences,35(6), 547-572. doi:10.1080/10408369891234273
[2] Hepatic lipase deficiency. (n.d.). Retrieved February 3, 2019, from https://rarediseases.info.nih.gov/diseases/12864/hepatic-lipase-deficiency
[3] Can triglycerides affect my heart health? (2018, September 13). Retrieved February 3, 2019, from https://www.mayoclinic.org/diseases-conditions/high-blood-cholesterol/in-depth/triglycerides/art-20048186
[4] High cholesterol. (2017, August 15). Retrieved February 3, 2019, from https://www.mayoclinic.org/diseases-conditions/high-blood-cholesterol/diagnosis-treatment/drc-20350806
[5] Hepatic lipase deficiency - Genetics Home Reference - NIH. (n.d.). Retrieved February 3, 2019, from https://ghr.nlm.nih.gov/condition/hepatic-lipase-deficiency#resources
[6] Wade, R. L., Andel, R. A., Rice, S. G., Banka, C. L., & Dyer, C. A. (2002). Hepatic Lipase Deficiency Attenuates Mouse Ovarian Progesterone Production Leading to Decreased Ovulation and Reduced Litter Size. Biology of Reproduction,66(4), 1076-1082. doi:10.1095/biolreprod66.4.1076
[7] Jacob, H. J. (1999). Functional genomics and rat models. Genome research, 9(11), 1013-1016.
[8] Grummer, R. R., & Carroll, D. J. (1988). A review of lipoprotein cholesterol metabolism: importance to ovarian function. Journal of animal science, 66(12), 3160-3173.
[9] Verhoeven, A.J., D. Carling, and H. Jansen, Hepatic lipase gene is transcribed in rat adrenals into a truncated mRNA. J Lipid Res, 1994. 35(6): p. 966-75.
[10] Verhoeven, A.J. and H. Jansen, Hepatic lipase mRNA is expressed in rat and human steroidogenic organs. Biochim Biophys Acta, 1994. 1211(1): p. 121-4.
[1] Connelly, P. W., & Hegele, R. A. (1998). Hepatic Lipase Deficiency. Critical Reviews in Clinical Laboratory Sciences,35(6), 547-572. doi:10.1080/10408369891234273
[2] Hepatic lipase deficiency. (n.d.). Retrieved February 3, 2019, from https://rarediseases.info.nih.gov/diseases/12864/hepatic-lipase-deficiency
[3] Can triglycerides affect my heart health? (2018, September 13). Retrieved February 3, 2019, from https://www.mayoclinic.org/diseases-conditions/high-blood-cholesterol/in-depth/triglycerides/art-20048186
[4] High cholesterol. (2017, August 15). Retrieved February 3, 2019, from https://www.mayoclinic.org/diseases-conditions/high-blood-cholesterol/diagnosis-treatment/drc-20350806
[5] Hepatic lipase deficiency - Genetics Home Reference - NIH. (n.d.). Retrieved February 3, 2019, from https://ghr.nlm.nih.gov/condition/hepatic-lipase-deficiency#resources
[6] Wade, R. L., Andel, R. A., Rice, S. G., Banka, C. L., & Dyer, C. A. (2002). Hepatic Lipase Deficiency Attenuates Mouse Ovarian Progesterone Production Leading to Decreased Ovulation and Reduced Litter Size. Biology of Reproduction,66(4), 1076-1082. doi:10.1095/biolreprod66.4.1076
[7] Jacob, H. J. (1999). Functional genomics and rat models. Genome research, 9(11), 1013-1016.
[8] Grummer, R. R., & Carroll, D. J. (1988). A review of lipoprotein cholesterol metabolism: importance to ovarian function. Journal of animal science, 66(12), 3160-3173.
[9] Verhoeven, A.J., D. Carling, and H. Jansen, Hepatic lipase gene is transcribed in rat adrenals into a truncated mRNA. J Lipid Res, 1994. 35(6): p. 966-75.
[10] Verhoeven, A.J. and H. Jansen, Hepatic lipase mRNA is expressed in rat and human steroidogenic organs. Biochim Biophys Acta, 1994. 1211(1): p. 121-4.
| thornton_finalpresentation_4-5-19_draft1.pptx | |
| File Size: | 3733 kb |
| File Type: | pptx |
| thornton_finalpresentation_5-2-19_draft2.pptx | |
| File Size: | 5371 kb |
| File Type: | pptx |